
Priothera Receives R&D Innovation Loan from Bpifrance
NEWS
April 2022
Priothera Receives R&D Innovation Loan from Bpifrance
€1.5 million loan will further support clinical manufacture of mocravimod for the adjunctive treatment of Acute Myeloid Leukemia (AML)
Dublin, Ireland and Saint-Louis, France – Monday 4 th April 2022 – Priothera, a late-clinical stage biotechnology company pioneering the development of its S1P receptor modulator drug, mocravimod, today announces that it has entered a €1.5 million Loan Agreement with the regional Bpifrance in Strasbourg (Grand Est Bpifrance), via Priothera SAS, its French affiliate.
This R&D innovation loan will be used to further support the clinical manufacture of mocravimod for a European, US and Asian registration-enabling clinical trial with mocravimod in Acute Myeloid Leukemia (AML) patients undergoing allogeneic hematopoietic stem cell transplant (HSCT).
Brice Suire, Co-Founder and Chief Financial Officer of Priothera, said: “This non-dilutive financing, alongside the funding from the European Investment Bank that we announced recently, will play an important role in financing the development of mocravimod. It will allow us to strengthen our local French team and accelerate delivery of the clinical supply of mocravimod needed for our upcoming registration-enabling global clinical trial. We are very pleased with the confidence shown by Grand Est Bpifrance in supporting the Company in its development program.”
Alban Stamm, Innovation Manager at Bpifrance Alsace commented: “We believe that Priothera, with its drug candidate mocravimod, has the potential to enable a major breakthrough in preventing transplant rejection which would provide a tremendous benefit to AML patients undergoing a stem cell transplant. The Bpifrance loan will support Priothera’s key objective of establishing human proof of concept and generating registrational data, allowing the company to create significant socio-economic value in France and beyond that for patients globally.”
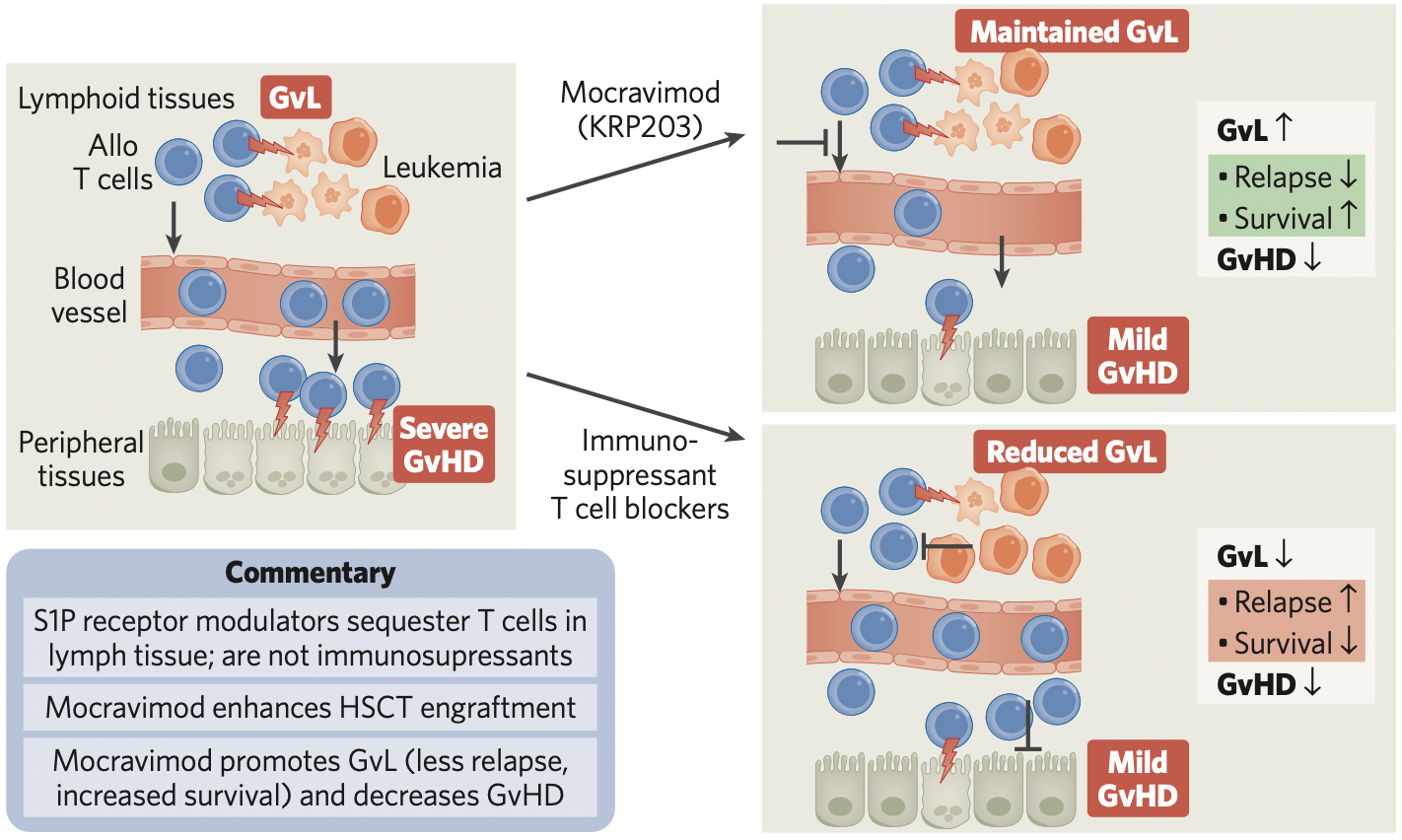
Mocravimod, a sphingosine 1 phosphate (S1P) receptor modulator which has been previously tested in multiple autoimmune indications, is being developed to enhance the curative potential of HSCT in patients with AML. Moreover, it has shown clinically relevant benefits in an early clinical study in patients with hematologic malignancies undergoing HSCT.
About Priothera
Priothera is leading the way in developing orally applied sphingosine 1 phosphate (S1P) receptor modulators for the treatment of hematological malignancies. S1P receptor modulators are known to largely reduce egress of T cells from lymphatic tissues and not being immunosuppressants, thereby allowing for inhibition of graft-versus-host-disease (GvHD) while maintaining graft-versus-leukemia benefits in patients receiving HSCT.
Priothera which was founded in 2020 by an experienced team of drug development experts is headquartered in Dublin, Ireland. The Company is backed by international founding investors Fountain Healthcare Partners (Dublin, Ireland), funds managed by Tekla Capital Management, LLC (Boston, Massachusetts), HealthCap (Stockholm, Sweden) and EarlyBird Venture Capital (Berlin, Germany).
For more information please visit: www.priothera.com
About Bpifrance
Bpifrance is the one stop shop for entrepreneurs with a vastly comprehensive toolbox offered in the field to customers through 50 local branches. Our mission is simple: we believe in serving the future, by being entrepreneur-centric and heavily decentralized.
Contact
Priothera
Philippe Lievre, Chief Business Officer Priothera
T: +41 76 802 11 30
E : philippe.lievre@priothera.com
MEDiSTRAVA Consulting
Sylvie Berrebi, David Dible, Sandi Greenwood, Frazer Hall
E: priothera@medistrava.com
T: +44 (0)7714 306525




Recent Comments